Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Micheau, C.; Ueda, Yuki; Motokawa, Ryuhei; Bauduin, P.*; Girard, L.*; Diat, O.*
Langmuir, 39(31), p.10965 - 10977, 2023/07
Times Cited Count:0 Percentile:0(Chemistry, Multidisciplinary)Kobayashi, Keita; Yamaguchi, Akiko; Okumura, Masahiko
Applied Clay Science, 228, p.106596_1 - 106596_11, 2022/10
Times Cited Count:6 Percentile:77.65(Chemistry, Physical)no abstracts in English
Okumura, Masahiko
Chikyu Kagaku, 55(4), p.110 - 121, 2021/12
no abstracts in English
Okamoto, Yoshihiro; Osugi, Takeshi; Shiwaku, Hideaki; Akabori, Mitsuo*
Insights Concerning the Fukushima Daiichi Nuclear Accident, Vol.4; Endeavors by Scientists, p.285 - 294, 2021/10
The transfer behavior of cesium adsorbed on some clay minerals in aqueous solution was investigated by X-ray absorption fine structure (XAFS) analysis of the Cs K-edge. The sample was prepared by mixing Cs-adsorbed mineral with a different pure clay mineral in water. The XAFS results of the dried mixture powder were compared with those obtained before mixing. It was recognized from the XAFS analysis for three kinds of clay minerals illite, kaolinite and vermiculite, that cesium was transferred from kaolinite to illite and vermiculite, and from illite to vermiculite. It can be concluded that cesium is transferred to and accumulated in vermiculite.
Fukuda, Tatsuya*; Takahashi, Ryo*; Hara, Takuhi*; Ohara, Koji*; Kato, Kazuo*; Matsumura, Daiju; Inaba, Yusuke*; Nakase, Masahiko*; Takeshita, Kenji*
Journal of Nuclear Science and Technology, 58(4), p.399 - 404, 2021/04
Times Cited Count:6 Percentile:60.71(Nuclear Science & Technology)Kimura, Tatsuki*; Kozai, Naofumi; Sakamoto, Fuminori; Fukutani, Satoshi*; Ikegami, Maiko*
Doboku Gakkai Rombunshu, G (Kankyo) (Internet), 76(7), p.III_375 - III_382, 2020/00
no abstracts in English
Yoshigoe, Akitaka
Hoshako Riyo No Tebiki, p.130 - 138, 2019/02
The educational book on next-generation synchrotron radiation usage is partially written. This book is being planned by several professors at Tohoku University. In this book, the contents which have already reported in our recent paper [Appl. Phys. Lett. 112 (2018) 021603.] are mainly written. The future perspective on photoemission electron microscopy using next generation synchrotron radiation and its possibility to analyze environmental samples and insulating functional materials are also described.
Okumura, Masahiko; Kerisit, S.*; Bourg, I. C.*; Lammers, L. N.*; Ikeda, Takashi*; Sassi, M.*; Rosso, K. M.*; Machida, Masahiko
Journal of Environmental Radioactivity, 189, p.135 - 145, 2018/09
Times Cited Count:51 Percentile:87.2(Environmental Sciences)no abstracts in English
Yoshigoe, Akitaka; Shiwaku, Hideaki; Kobayashi, Toru; Shimoyama, Iwao; Matsumura, Daiju; Tsuji, Takuya; Nishihata, Yasuo; Kogure, Toshihiro*; Okochi, Takuo*; Yasui, Akira*; et al.
Applied Physics Letters, 112(2), p.021603_1 - 021603_5, 2018/01
Times Cited Count:6 Percentile:34.59(Physics, Applied)A synchrotron radiation photoemission electron microscope (SR-PEEM) was applied to demonstrate pinpoint analysis of micrometer-sized weathered biotite clay particles with artificially adsorbed cesium (Cs) atoms. Despite the insulating properties of the clay, we observed the spatial distributions of constituent elements (Si, Al, Cs, Mg, Fe) without charging issues. We found that Cs atoms were likely to be adsorbed evenly over the entire particle. Spatially-resolved X-ray absorption spectra (XAS) of the Cs M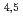 -edge region showed Cs to be present in a monocation state (Cs
-edge region showed Cs to be present in a monocation state (Cs ). Further pinpoint XAS measurements were also performed at the Fe L
). Further pinpoint XAS measurements were also performed at the Fe L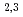 -edge to determine the chemical valence of the Fe atoms. Our results demonstrate the utility of SR-PEEM as a tool for spatially-resolved chemical analyses of various environmental substances, which is not limited by the poor conductivity of samples.
-edge to determine the chemical valence of the Fe atoms. Our results demonstrate the utility of SR-PEEM as a tool for spatially-resolved chemical analyses of various environmental substances, which is not limited by the poor conductivity of samples.
Sassi, M.*; Rosso, K. M.*; Okumura, Masahiko; Machida, Masahiko
Clays and Clay Minerals, 65(5), p.371 - 375, 2017/11
Times Cited Count:2 Percentile:79.17(Chemistry, Physical)no abstracts in English
Araki, Yuki*; Sato, Hisao*; Okumura, Masahiko; Onishi, Hiroshi*
Surface Science, 665, p.32 - 36, 2017/11
Times Cited Count:11 Percentile:48.83(Chemistry, Physical)no abstracts in English
Sassi, M.*; Okumura, Masahiko; Machida, Masahiko; Rosso, K. M.*
Physical Chemistry Chemical Physics, 19(39), p.27007 - 27014, 2017/10
Times Cited Count:4 Percentile:17(Chemistry, Physical)no abstracts in English
Okumura, Masahiko; Sassi, M.*; Rosso, K. M.*; Machida, Masahiko
AIP Advances (Internet), 7(5), p.055211_1 - 055211_9, 2017/05
Times Cited Count:6 Percentile:29.59(Nanoscience & Nanotechnology)no abstracts in English
Lammers, L.*; Bourg, I. C.*; Okumura, Masahiko; Kolluri, K.*; Sposito, G.*; Machida, Masahiko
Journal of Colloid and Interface Science, 490, p.608 - 620, 2017/03
Times Cited Count:107 Percentile:93.33(Chemistry, Physical)no abstracts in English
Kerisit, S.*; Okumura, Masahiko; Rosso, K. M.*; Machida, Masahiko
Clays and Clay Minerals, 64(4), p.389 - 400, 2016/08
Times Cited Count:29 Percentile:74.44(Chemistry, Physical)no abstracts in English
Sassi, M.*; Rosso, K. M.*; Okumura, Masahiko; Machida, Masahiko
Clays and Clay Minerals, 64(2), p.108 - 114, 2016/04
Times Cited Count:6 Percentile:21(Chemistry, Physical)no abstracts in English
Machida, Masahiko; Okumura, Masahiko; Nakamura, Hiroki; Sakuramoto, Kazuhiro*
Proceedings of Joint International Conference on Mathematics and Computation, Supercomputing in Nuclear Applications and the Monte Carlo Method (M&C + SNA + MC 2015) (CD-ROM), 12 Pages, 2015/04
In order to clarify physicochemical behaviors of radioactive Cs released into environment from the Fukushima Daiichi Nuclear Power Plants, we study on two issues, i.e., hydration structures of Cs and its adsorption on a specific edge in a clay particle (mica) by employing first principles calculations. In this paper, firstly, we report on hydration structures of Cs
and its adsorption on a specific edge in a clay particle (mica) by employing first principles calculations. In this paper, firstly, we report on hydration structures of Cs by using Born-Oppenheimer first-principles molecular dynamics. Our striking finding in the hydration structures is that Cs
by using Born-Oppenheimer first-principles molecular dynamics. Our striking finding in the hydration structures is that Cs has no clear second hydration shell in contrast to any other alkali cations. Secondly, we construct a model of the Frayed Edge Site and confirm that the model actually becomes selective for Cs when expanding the interlayer distance from that of the original crystal structure through the calculation of the ion-exchange energy.
has no clear second hydration shell in contrast to any other alkali cations. Secondly, we construct a model of the Frayed Edge Site and confirm that the model actually becomes selective for Cs when expanding the interlayer distance from that of the original crystal structure through the calculation of the ion-exchange energy.
Matsumura, Daiju; Kobayashi, Toru; Miyazaki, Yuji; Okajima, Yuka*; Nishihata, Yasuo; Yaita, Tsuyoshi
Clay Science, 18(4), p.99 - 105, 2014/12
Yaita, Tsuyoshi; Kobayashi, Toru; Ikeda, Takashi; Matsumura, Daiju; Machida, Masahiko; Okumura, Masahiko; Nakamura, Hiroki
Hoshako, 27(6), p.315 - 322, 2014/11
no abstracts in English
Shimoyama, Iwao; Hirao, Norie; Baba, Yuji; Izumi, Toshinori; Okamoto, Yoshihiro; Yaita, Tsuyoshi; Suzuki, Shinichi
Clay Science, 18(3), p.71 - 77, 2014/09
no abstracts in English